 
it's dead-on, and all I did was use #Z# as a correctional factor in the ideal gas law. Molar Masses NaHCO3:84.006609 g/mol CO2:44.0095 g/mol Mole Ratio From the balanced equation, the mole ratio between NaHCO3 and CO2 is 3 mol NaHCO3:3 mol CO2. Since #Z < 1#, #"CO"_2# is easier to compress than a comparable ideal gas (thus its molar volume is less than #22.711# at #"1 bar"# and #"273.15 K"#). If #Z = 1#, then #"CO"_2# is perfectly ideal.įrom this website again, I get #Z = 0.9934#. To convert from the usual ppmv units to ppm mass, multiply by the ratio of the molar weight of CO 2 to that of air, i.e. 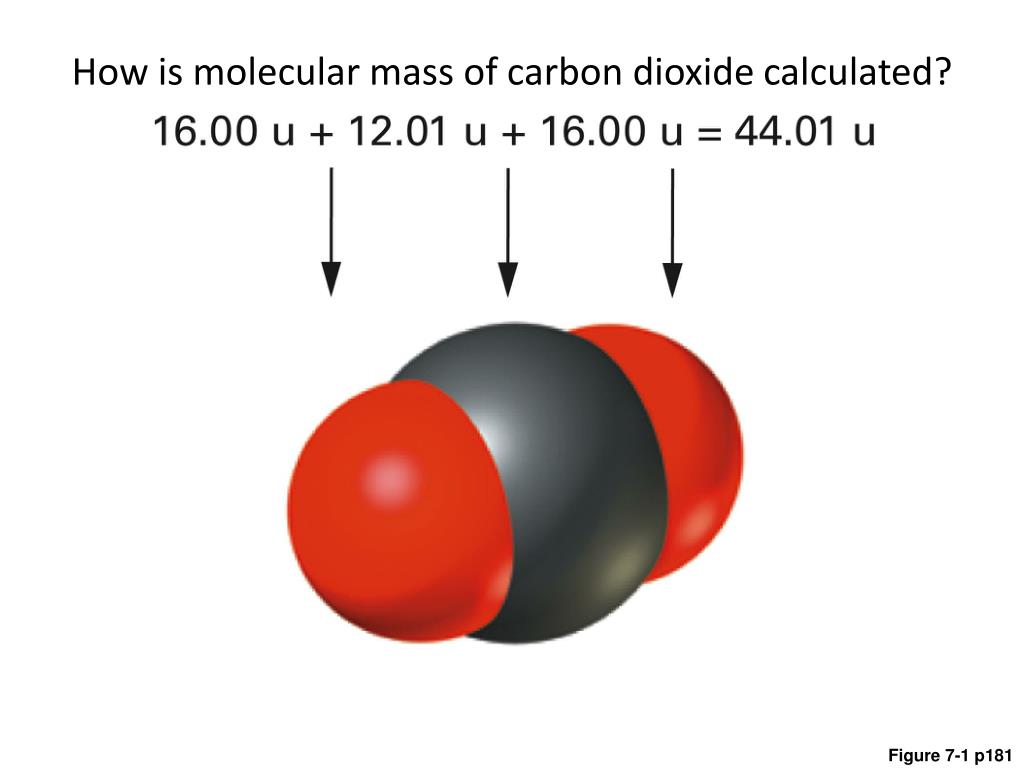
We can also use the compressibility factor #Z = (PV)/(nRT)#, which is an empirical constant related to how easily #"CO"_2# responds to compression. Carbon dioxide concentrations have varied widely over the Earths 4.54 billion year history. That is about #0.72%# error from the true density, which is quite good. The molar mass of a substance tells you how much one mole of that substances. #R# is the universal gas constant, #"0.083145 L"cdot"bar/mol"cdot"K"#. So, the molar mass of carbon dioxide is 44.01 g/mol.How we calculate mass from moles Mass of any substance will be calculated from moles as. STP currently involves #"1 bar"# pressure. 6.15 grams of carbon dioxide is form when 8.94 g of oxygen is completely react. To get an idea of how the density is like when assuming ideality, we can use the ideal gas law to compare. This also gives a real mass density of #color(blue)("0.001951 g/mL")# at #"1 bar"# and "C"#. 
Or, one can calculate this from this website. Carbon dioxide Molecular Formula A chemical formula is a way to communicate information in chemistry about the proportions of atoms that make up a specific chemical compound or molecule. Element/Moleculeīy knowing the molar mass of a substance, having any mass of that substance allows one to know how many moles of it they have.Wikipedia gives the density as #"0.001977 g/mL"# at #"1 atm"#, or if we convert it for #"1 bar"#, #color(blue)("0.001951 g/mL")#. Molar Mass of Carbon dioxide (CO 2) is 44.01 g/mol. A list of some molar masses can be seen below. 1 gm atom of 12C 12.0 g 6.022 × 1023 atoms of 12Carbon weigh 12.0 g 12 1 atom of 12 Carbon will weigh or or 36 Ar 38 Ar 40 Ar Ans. We can use the Ideal Gas Law to determine its density. The molar mass of any element or molecule is given by the sum of the atomic weights multiplied by 1 g/mol. Explanation: The molar mass of CO2 is 0.044 kg/mol. In SI units, the molar mass is given by kg/ mol, however by convention the molar mass is expressed in units of g/mol. We assume you are converting between grams CO2 and mole.You can view more details on each measurement unit: molecular weight of CO2 or mol This compound is also known as Carbon Dioxide.The SI base unit for amount of substance is the mole. How many grams CO2 in 1 mol The answer is 44.0095. Also, important in this field is Avogadros. This comparison is seen below:ġ dozen = 12 objects 1 mol = 6.022141 x 10 23 objects More information from the unit converter. It is defined to be 1/12 of the mass of one atom of carbon-12 and in older works is also abbreviated as amu. However when talking about moles, instead of only having a dozen, there is in fact 6.022141 x 10 23 of a given substance in a mole (known as Avogadro's number). For example, if someone has a dozen bricks and another person has a dozen feathers, they have the same amount of their substance, but a vastly different total mass. Since a mole is defined as the amount of a substance and substances have different masses, each element or molecule will have a different molar mass. Molar mass (M) is a physical property, defined as the mass of a given element or molecule per mole of that substance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
